Research & Development
Sealife Pharma is working hard to produce a new generation of first in class small molecule antibiotic drugs to negate and overcome the issue of multi drug resistant bacteria.
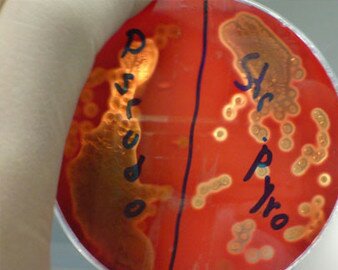
Sealife’s antibiotic discovery program was initiated in 2008 and focused on the isolation of compounds produced by the fermentation of marine fungi. Lead structure isolation, testing and characterization in different screening and analytical platforms were then performed to isolate compounds of interest for further SAR. Sealife used more than 90 different clinically relevant bacterial and fungal strains covering nearly all clinical indications as well as 6 different groups of viruses, with a strong focus on skin and respiratory
tract infections.
The internal screening process is regulated by European standards according to EUCAST (=European Committee for Antimicrobial Susceptibility Testing) and AFST (= Antifungal Susceptibility Testing) criteria based on the European Society of Clinical Microbiology and Infectious Diseases (ESCMID).
Additionally to the activity screening, a broad toxicological screening platform in HTS was established to check the general toxicity (Alamar blue, AnnexinV/PI, Caspase, MTT-Assay, HERG-Test) and the genotox/mutagenic (HPRT Assay, Comet Assay, Micronucleus Assay, Mitotic index) activity of compounds.
Tests were done in 2D and innovative 3D systems including human primary cells.
Potential drug candidates have been selected on the basis of certain minimum standards of biological activity, reproducibility, economical production, broad spectrum of activity and acceptable toxic profile.
After HIT identification SLP’s in-house chemical synthesis platform developed a synthesis protocol for the
lead structures based on medical chemistry considerations and cost-efficiency.
The process has led to two substance-programs (SLP0904 and SLP0905) that are currently in further development and an additional pipeline of promising candidates in the Gram-negative field.